DDX41 was identified as a causative gene for late-onset familial myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML). While DDX41 is thought to be one of the most frequent targets of germline mutations responsible for sporadic cases with AML/MDS and other myeloid neoplasms, the entire spectrum of pathogenic DDX41 variants and their effect size therein are still to be elucidated, and so was the clinical picture of DDX41-mutated myeloid neoplasms.
In this study, through an international collaboration, we investigated DDX41 variants in a total of 5,609 sporadic cases with different myeloid neoplasms from different ethnicities, using next generation sequencing. Mutations in the major driver genes commonly mutated in AML/MDS were also examined. Frequencies of germline DDX41 variants were compared between sporadic cases with myeloid neoplasms and healthy individuals (n=13,906). We also characterized genetic/clinical features of DDX41-mutated myeloid neoplasms.
We identified a total of 208 (3.6%) patients with DDX41 variants, of whom approximately 50% had both germline and somatic mutations, whereas 37% and 13% had either germline or somatic mutations alone, respectively. Somatic mutations were found in 58% of patients with germline mutation, which was significantly higher than those without (0.21%) (P<0.0001). No somatic mutation was identified in healthy individuals. Among 174 germline variants, truncating and missense mutations were found in 93 and 81 cases, respectively, whereas only 1.9% of somatic mutations were truncating (P<0.0001). Among 21 cases with somatic mutations alone, 4 had multiple somatic mutations and an additional 4 had loss-of-heterozygosity of the DDX41 locus (5q35.3), including 3 with uniparental disomy and 1 with deletion. Thus, 8 out of 21 cases with somatic mutation alone were suspected to have biallelic DDX41 mutations.
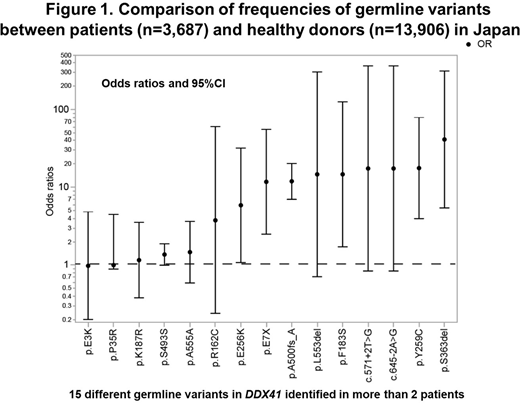
Germline DDX41 variants showed a conspicuous ethnic diversity; the most frequent germline variants were A500fs in Japan, D140fs in USA, Q41* in Germany, G218D in Italy, M1I in Sweden, S21fs in Thailand. The M1I variant was also seen in other European countries, but not in Japan or Thailand, while no A500fs mutation was found in Europe. Among the Japanese population, significant enrichment in myeloid neoplasms was observed not only for truncating variants, such as A500fs (odds ratio (OR)=12.1) and E7X (OR=11.0) but also for missense variants, including Y259C (OR 14.3) and E256K (OR 7.81), frequently accompanied by a somatic DDX41 mutation (Figure 1).
Patients with germline and/or somatic DDX41 variants were significantly older than those without (P=0.00076) and more prevalent in male than female (OR=3.14; P<0.0001). DDX41 variants were significantly more frequent in MDS (4.7%) and AML (2.9%), compared with other myeloid neoplasms (0.58%). Among AML, mutations were more frequent in AML with myelodysplasia-related changes (P<0.00001). Patients with MDS having both germline and somatic mutations were more likely to classified in refractory anemia with excess blasts (RAEB), compared with those with germline or somatic alone (P=0.029). DDX41 variants were significantly associated with lower WBC and granulocyte counts. Most frequently co-occurring mutations included those in ASXL1, SRSF2, TET2, CUX1, and DNMT3A, of which only CUX1 mutations were statistically significant. Overall, no difference was observed in overall survival (OS) between DDX41-mutated and unmutated cases. However, among RAEB cases, DDX41 variants were associated with a significantly longer OS (P=0.0039).
In summary, the majority of DDX41-mutated cases had a germline variant, although a minority had somatic mutations alone. Pathogenic DDX41 alleles have a large ethnic diversity, where not only truncating variants but also missense variants are associated with an increased risk of the development of myeloid neoplasms.
Kanda:Chugai Pharma: Honoraria, Research Funding; Merck Sharp & Dohme: Honoraria; Mundipharma: Honoraria; Ono Pharmaceutical: Honoraria; Nippon Shinyaku: Honoraria, Research Funding; Takeda Pharmaceuticals: Honoraria; Alexion Pharmaceuticals: Honoraria; Shire: Honoraria; Mochida Pharmaceutical: Honoraria; Daiichi Sankyo: Honoraria; Shionogi: Research Funding; Meiji Seika Kaisha: Honoraria; Sanofi: Honoraria, Research Funding; Otsuka: Honoraria, Research Funding; Janssen: Honoraria; Pfizer: Honoraria, Research Funding; Eisai: Honoraria, Research Funding; Bristol-Myers Squibb: Honoraria; Celgene: Honoraria; Sumitomo Dainippon Pharma: Honoraria; Novartis: Honoraria; Kyowa Kirin: Honoraria, Research Funding; Astellas Pharma: Honoraria, Research Funding. Miyazaki:NIPPON SHINYAKU CO.,LTD.: Honoraria; Sumitomo Dainippon Pharma Co., Ltd.: Honoraria; Kyowa Kirin Co., Ltd.: Honoraria; Novartis Pharma KK: Honoraria; Astellas Pharma Inc.: Honoraria; Otsuka Pharmaceutical: Honoraria; Chugai Pharmaceutical Co., Ltd.: Honoraria; Celgene: Honoraria. Maciejewski:Alexion, BMS: Speakers Bureau; Novartis, Roche: Consultancy, Honoraria. Ogawa:Otsuka Pharmaceutical Co., Ltd.: Research Funding; Sumitomo Dainippon Pharma Co., Ltd.: Research Funding; KAN Research Institute, Inc.: Membership on an entity's Board of Directors or advisory committees, Research Funding; Asahi Genomics Co., Ltd.: Current equity holder in private company; Chordia Therapeutics, Inc.: Membership on an entity's Board of Directors or advisory committees, Research Funding; Eisai Co., Ltd.: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal